Why Pharma Should Rethink How To Use Social Media
 Last Thursday, I had the great fortune to address a group of prestigious individuals at the American College of Cardiology Professional and Corporate Consortium in Washington, D.C., on the relevant topic of Digital Health and Patient Engagement. The audience was primarily made up of professionals from pharmaceutical companies and medical device businesses, and as I spoke on the social media challenges these industries face when adopting digital and social initiatives, an ongoing discussion began centering primarily around the hurdles of regulatory requirements and constraints to reach patients directly.
Last Thursday, I had the great fortune to address a group of prestigious individuals at the American College of Cardiology Professional and Corporate Consortium in Washington, D.C., on the relevant topic of Digital Health and Patient Engagement. The audience was primarily made up of professionals from pharmaceutical companies and medical device businesses, and as I spoke on the social media challenges these industries face when adopting digital and social initiatives, an ongoing discussion began centering primarily around the hurdles of regulatory requirements and constraints to reach patients directly.
Getting down to it, the impact of patients and practitioners going online to research, communicate and make healthcare decisions has transformed the entire healthcare communications paradigm. For the pharmaceutical industry, this shift has been met with trepidation of how to effectively embrace the opportunity of leveraging social media for business goals. As noted in the ACC meeting, fear of regulatory compliance (and the lack of clear, cohesive guidance from the Food and Drug Administration), privacy concerns, and simply the unfamiliarity of social media – has kept pharma out of playing a hand at the crowded social media table. Further, according to the January 2014 report from the IMS Health Institute, almost 2/3 of respondents in the DHC/Google Executive Landscape 2013 survey agreed that the pharma/device industry is very far behind other industries with respect to the use of social media.
 This advent of transparent, real time public communications with consumers and healthcare providers (and at the behest from the industry requesting clarity on internet/social promotions) led the FDA to finally develop directional guidances, which were passed down in June 2014. The proposal addressed promotions posted with character space limitations (such as Twitter and Google/Bing search ads) must present a fair and balanced benefit and risk and include a direct link to more detailed risks. The FDA also discussed how companies can correct misinformation on third party sites, like Wikipedia and chat forums, as long as the information is accurate, clearly defined and non misleading. (Wikipedia is the leading single source of healthcare information for patients and healthcare professionals).
This advent of transparent, real time public communications with consumers and healthcare providers (and at the behest from the industry requesting clarity on internet/social promotions) led the FDA to finally develop directional guidances, which were passed down in June 2014. The proposal addressed promotions posted with character space limitations (such as Twitter and Google/Bing search ads) must present a fair and balanced benefit and risk and include a direct link to more detailed risks. The FDA also discussed how companies can correct misinformation on third party sites, like Wikipedia and chat forums, as long as the information is accurate, clearly defined and non misleading. (Wikipedia is the leading single source of healthcare information for patients and healthcare professionals).
However, with this piecemeal approach from the FDA, the guidance’s will likely do little to change any immediate behaviors of pharma companies on social media.
Another challenge is a question that we hear from all clients who are new to social media at one point or another: How can you truly quantify social media ROI? For pharma, the IMS Institute suggests they take a more refined approach – “Social media ROI requires a more nuanced view of the benefit of customer interaction, including the quantity and quality of followers, the number of comments, likes and shares, the reach to targeted demographics, the sentiment of comments and buzz, and ultimately the change in brand perception.” In this lens, providing content that fosters community involvement, thought leadership and purposeful messaging for a wide-spread audience could largely change the way customers interacted with these pharma brands.
As Pew Research indicates, 72% of adult internet users have searched online for information about health issues (the most popular being specific diseases and treatments). The pharmaceutical industry should consider it an obligation to provide meaningful education content to help accurately inform consumers about health related issues and out shine any misleading information that is on the world wide web. I would not dare suggest that pharma should axe promotions altogether, rather shift from promoting products as the core social media focus to instead developing a strategy that empowers their customers with critical and up-to-date health information.
Especially for pharma companies with a consumer products side to their business, activating strategies that provide significant content to practitioners and consumer would fall in line with the extremely successful social media communities of their healthcare cousins – Mayo Clinic and Cleveland Clinic. As the social achievements of those two social media juggernauts show, social media should be about creating messaging for that can help their audience, and not to serve as a mega phone for brand promotions. Take Johnson & Johnson for example – they were ranked 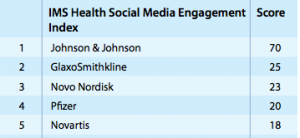 number one by a wide margin in the IMS Institute report of the top ten pharmaceutical companies by social media engagement. Digging deeper, Johnson & Johnson primarily focuses on non-prescription consumer products for their social strategy, but they are setting new precedents in other ways as well. They are one of the very few pharma companies that allows comments on their YouTube channel! On the non-consumer product side (and number three on this list) Novo Nordisk uses Pinterest to share visual information that practitioners can share as critical information pieces for families of children who have been recently diagnosed with diabetes.
number one by a wide margin in the IMS Institute report of the top ten pharmaceutical companies by social media engagement. Digging deeper, Johnson & Johnson primarily focuses on non-prescription consumer products for their social strategy, but they are setting new precedents in other ways as well. They are one of the very few pharma companies that allows comments on their YouTube channel! On the non-consumer product side (and number three on this list) Novo Nordisk uses Pinterest to share visual information that practitioners can share as critical information pieces for families of children who have been recently diagnosed with diabetes.
“We see that pharmaceutical companies recognize that patients increasingly are using some form of social media to obtain information, exchange views and seek advice regarding their healthcare. They therefore need to be present, active and engaged in these channels to be patient-centered.” Murray Aitken – Executive Director, IMS Institute for Healthcare Informatics.
Although uncertain if any remaining directives from the FDA would change the reluctancy of social adoption, changing the focus from brand-centric marketing to customer-centric marketing would alleviate some compliance fears and perhaps provide a more viable long-term approach to social media. So stick your toes in, Pharma … it’s not as cold as you think.
About Emily Trayers
Emily is the COO and SVP of Business Development with SayItSocial, and handles all key accounts while driving the acquisition and support of new clients. She is also one of our lead trainers and speaks at nationwide conferences and panels on the subject of social and digital business.

Like What You Are Reading? Subscribe To Read More
Join our mailing list to receive the latest news and updates from our team.